Lc Chemistry Notes
Notes tagged with "lc chemistry".
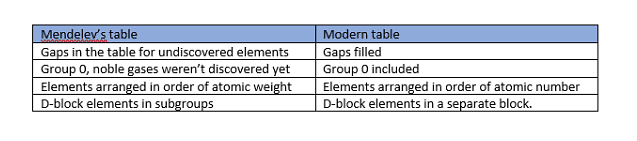
Periodic Table
Here are some differences between Mendeleev's Periodic Table and the Modern Periodic Table
Chemical Bonding Notes
- Compound: two or more different elements combined together chemically - Octet Rule: when bonding occurs, atoms tend to reach an electron arrangement with 8 electrons in the outermost energ...
The Periodic Table Groups
A group (also known as a family) is a vertical column in the periodic table of elements.The alkali metals – Group I - Very reactive soft metals. - Increases in reactivity down the group. - ...
Organic Chemistry – Fuels & Heats of Reaction
Organic chemistry is the chemistry of carbon containing compounds. A hydrocarbon is a substance that contains only carbon and hydrogen. There are three main hydrocarbons on the course – al...
Organic Compounds – Tetrahedral & Planar carbons
Tetrahedral carbon compounds In saturated organic compounds, all of the carbon atoms are tetrahedral.Chloroalkanes - A chloroalkane is an alkane in which one or more hydrogen atoms are rep...
Experiment: To prepare ethyne and examine its properties
Ethyne gas is prepared by the reaction of water with calcium carbide (CaC2). Ethyne gas is a colourless gas.Main Chemicals required - Calcium Carbide (CaC2) [also known as calcium dicarbide...